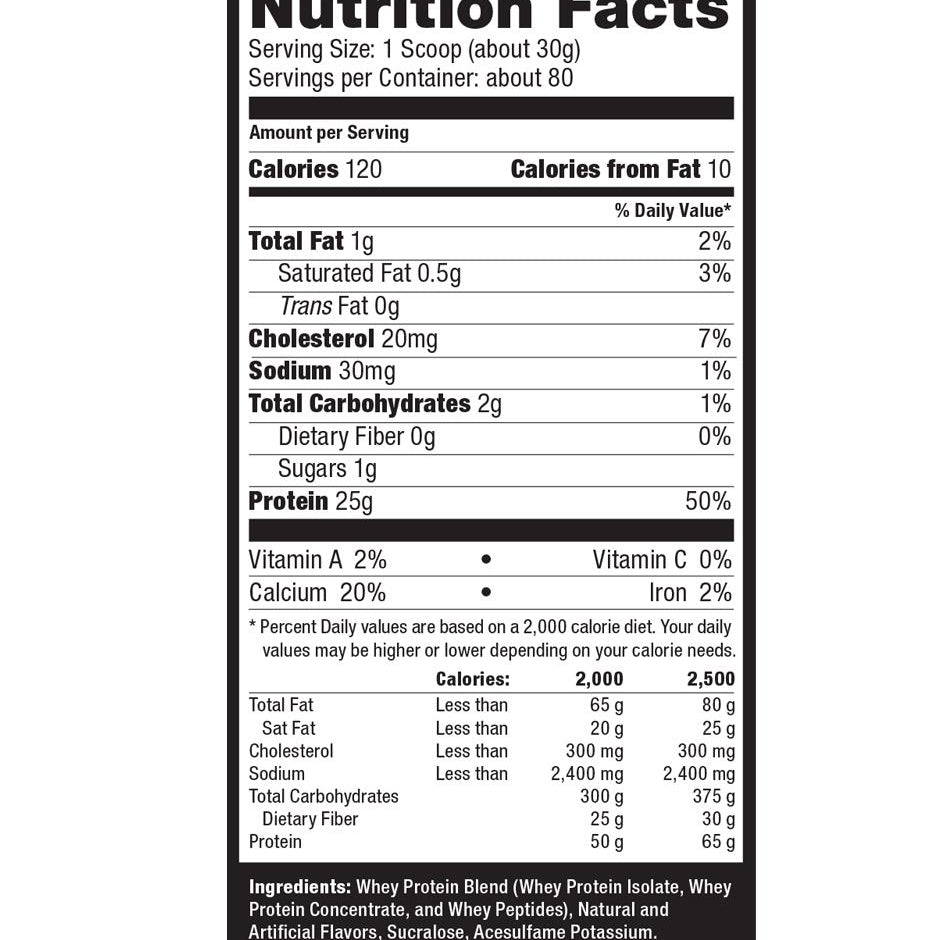
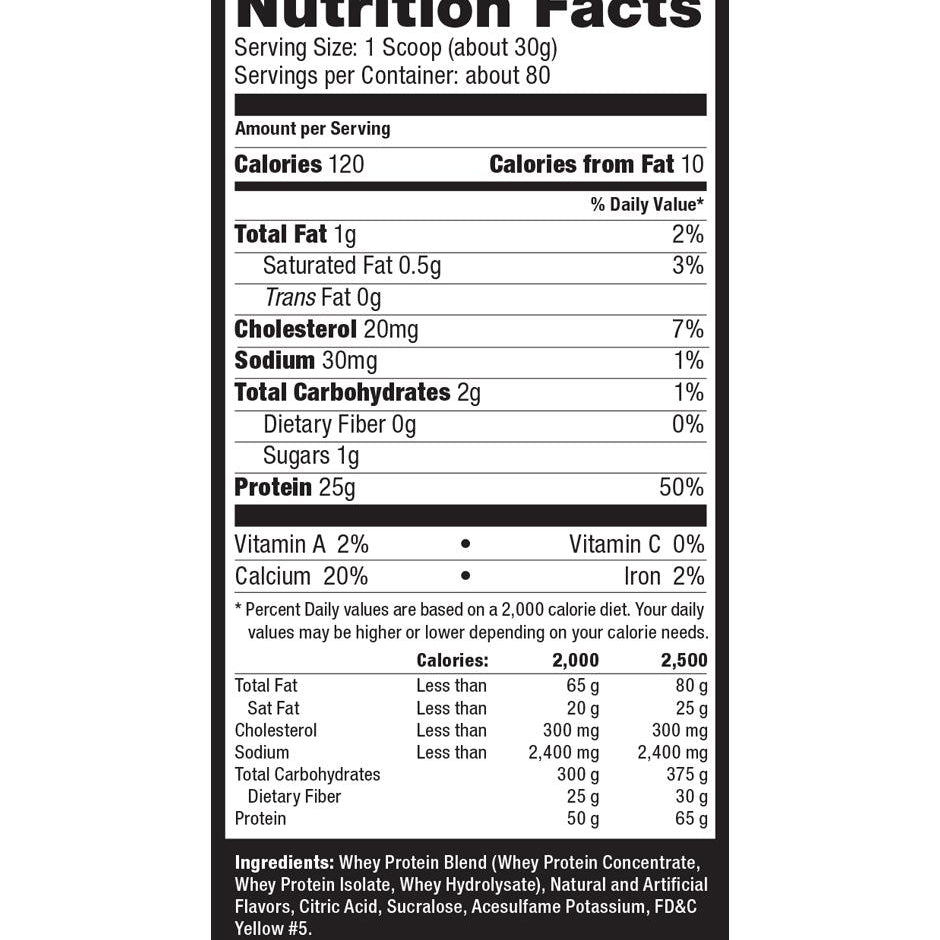
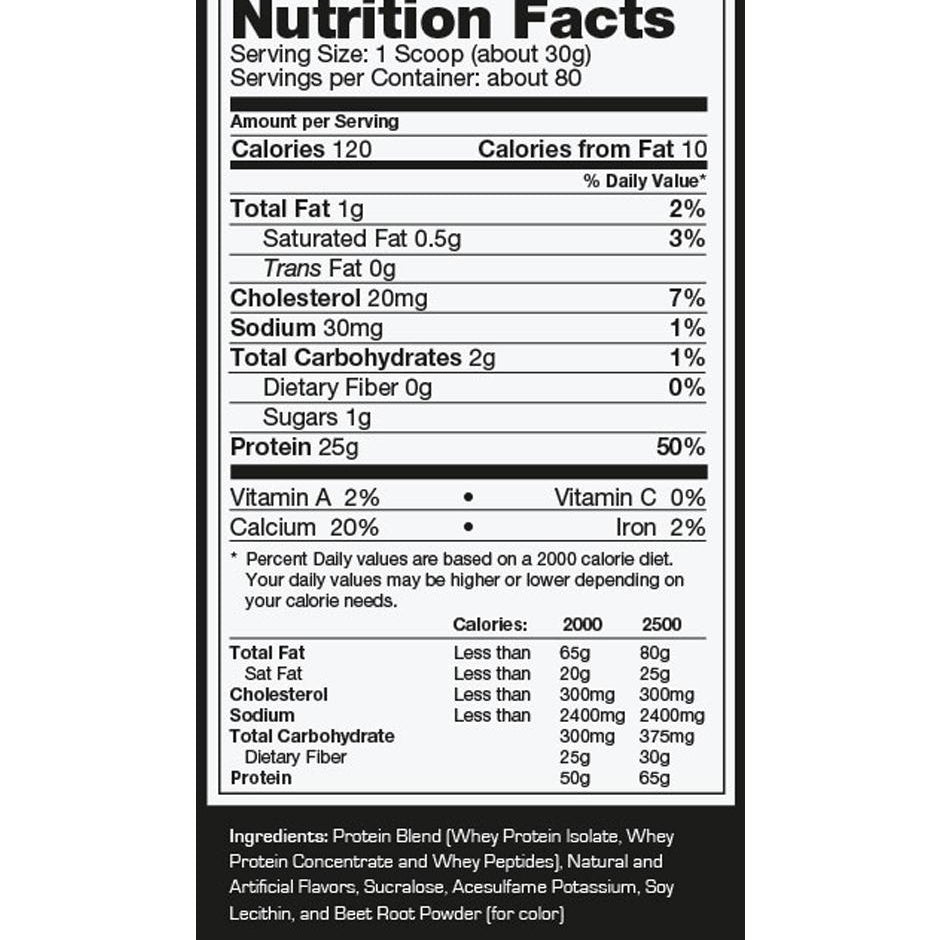
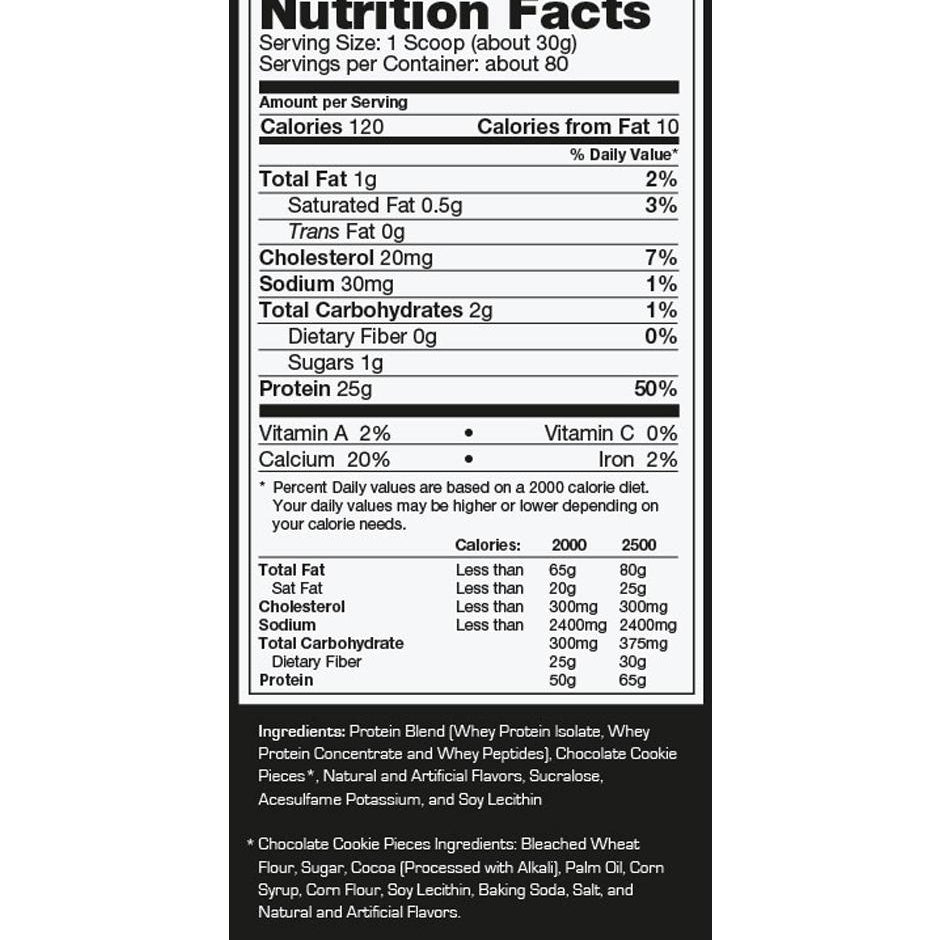
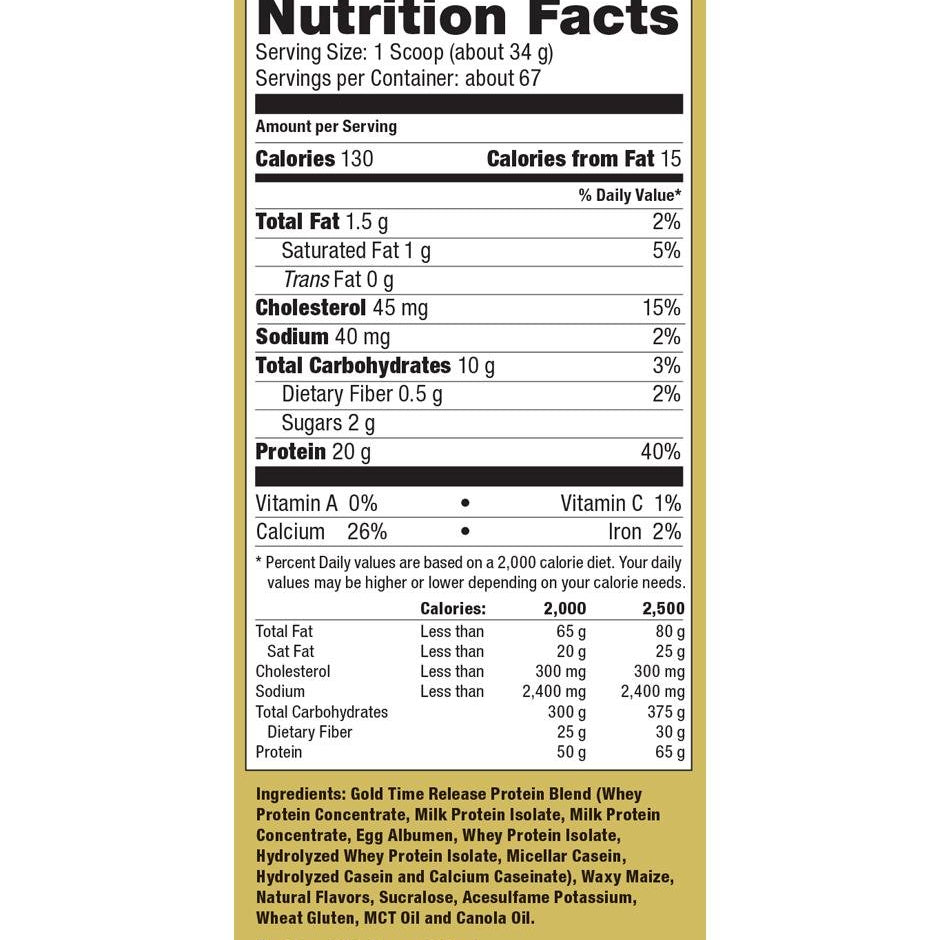
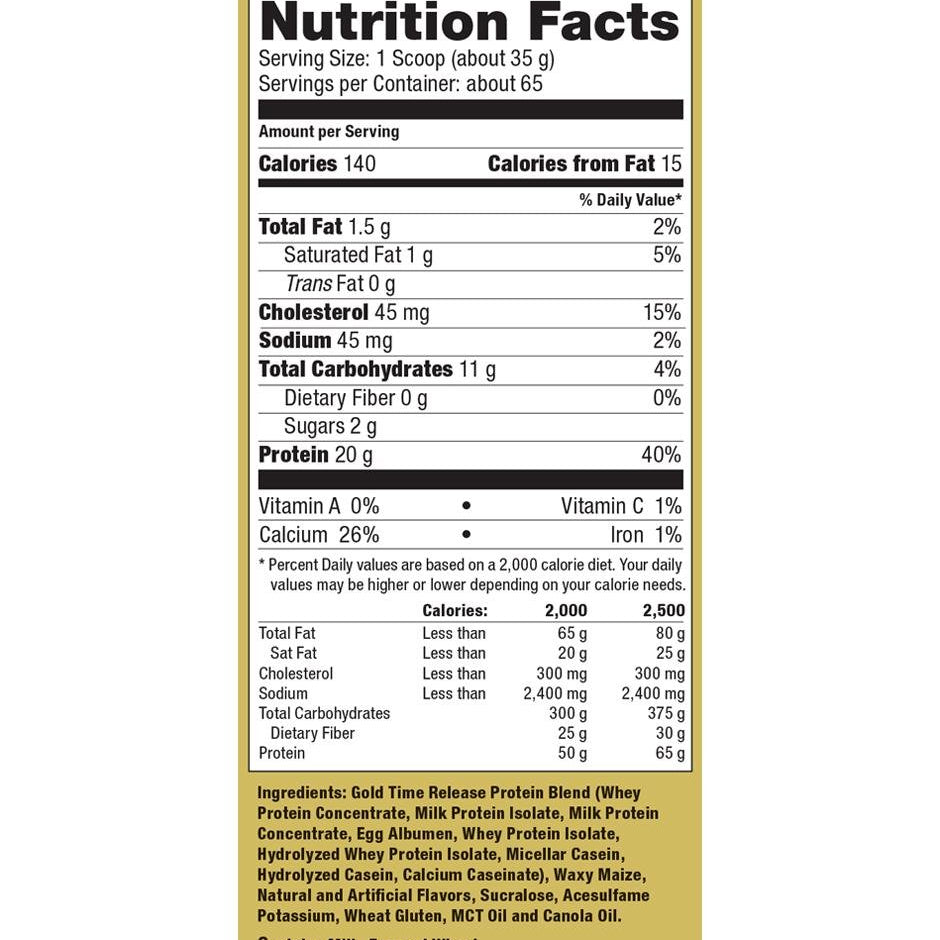
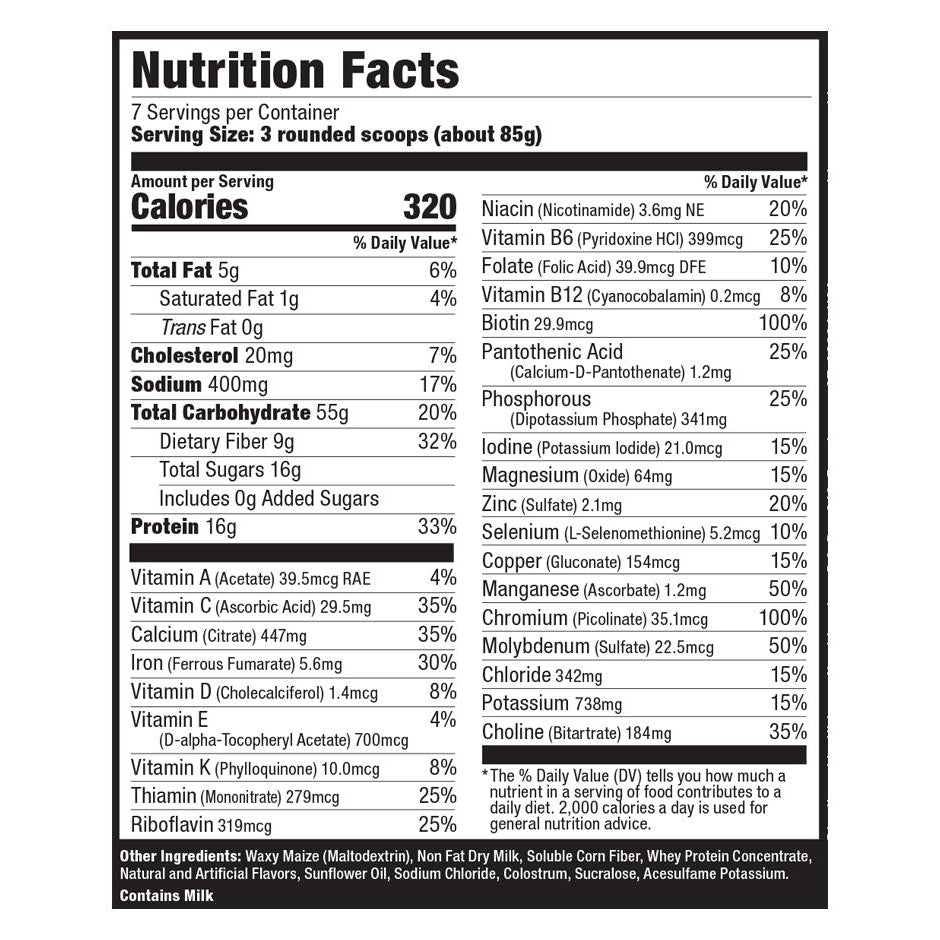
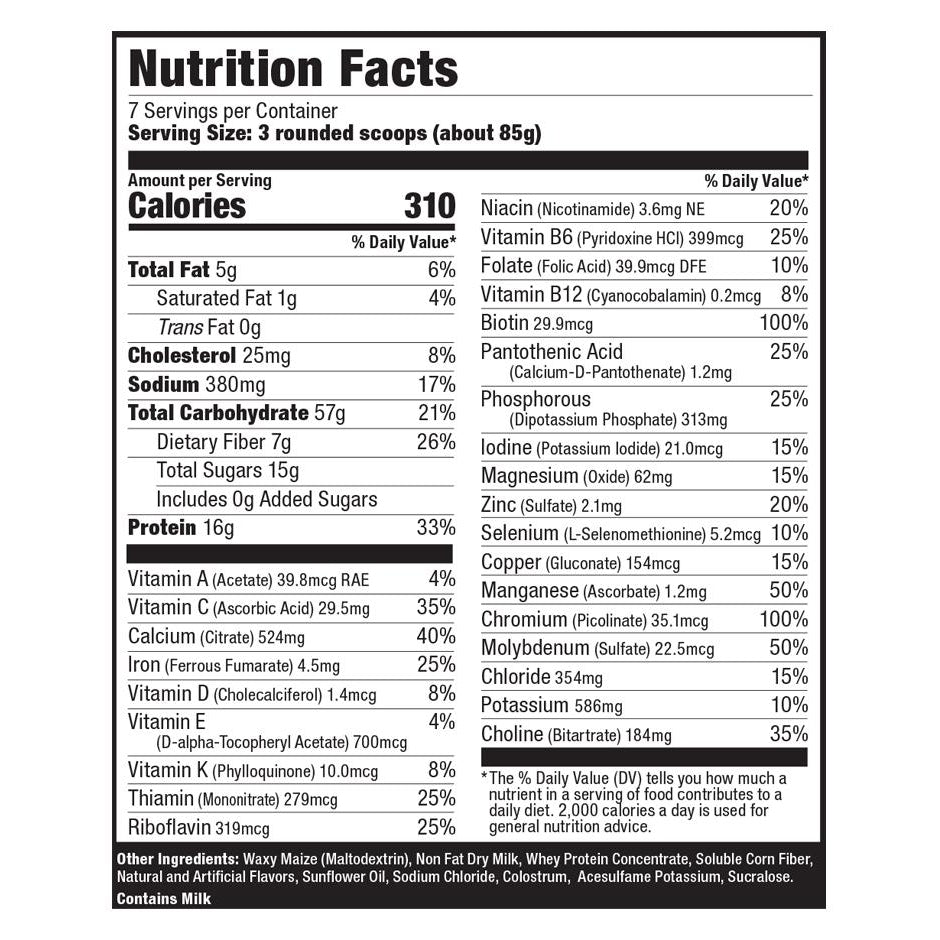
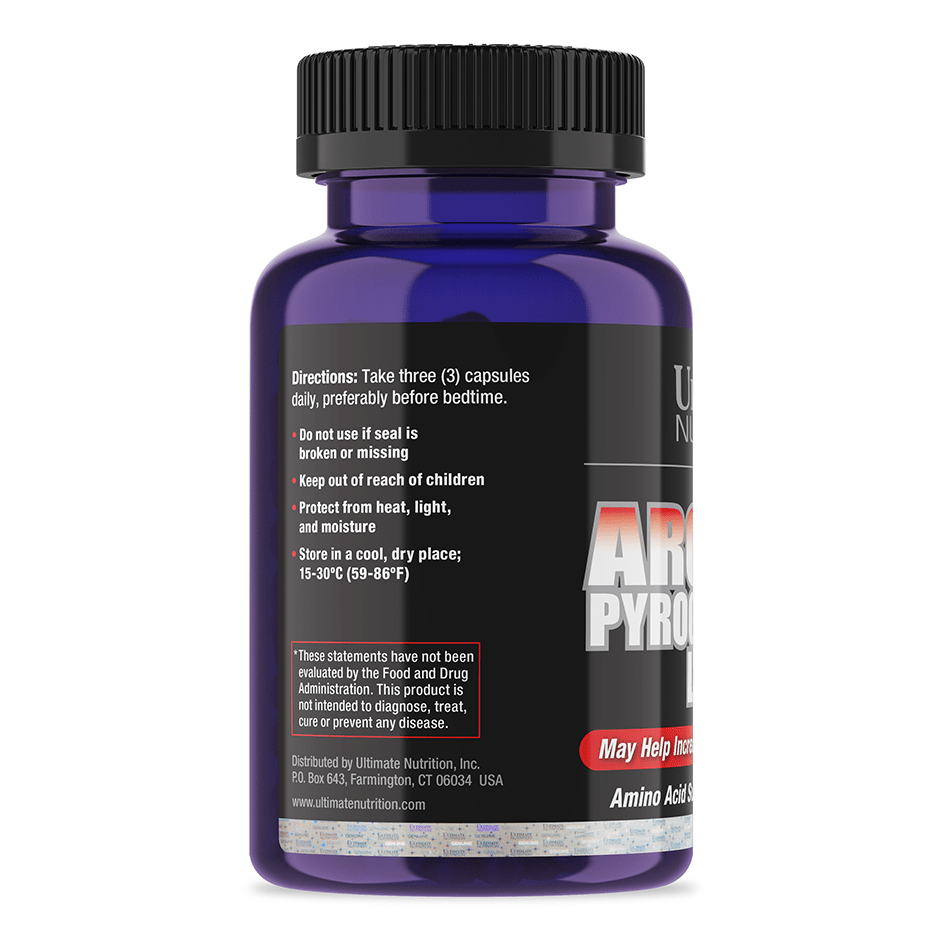
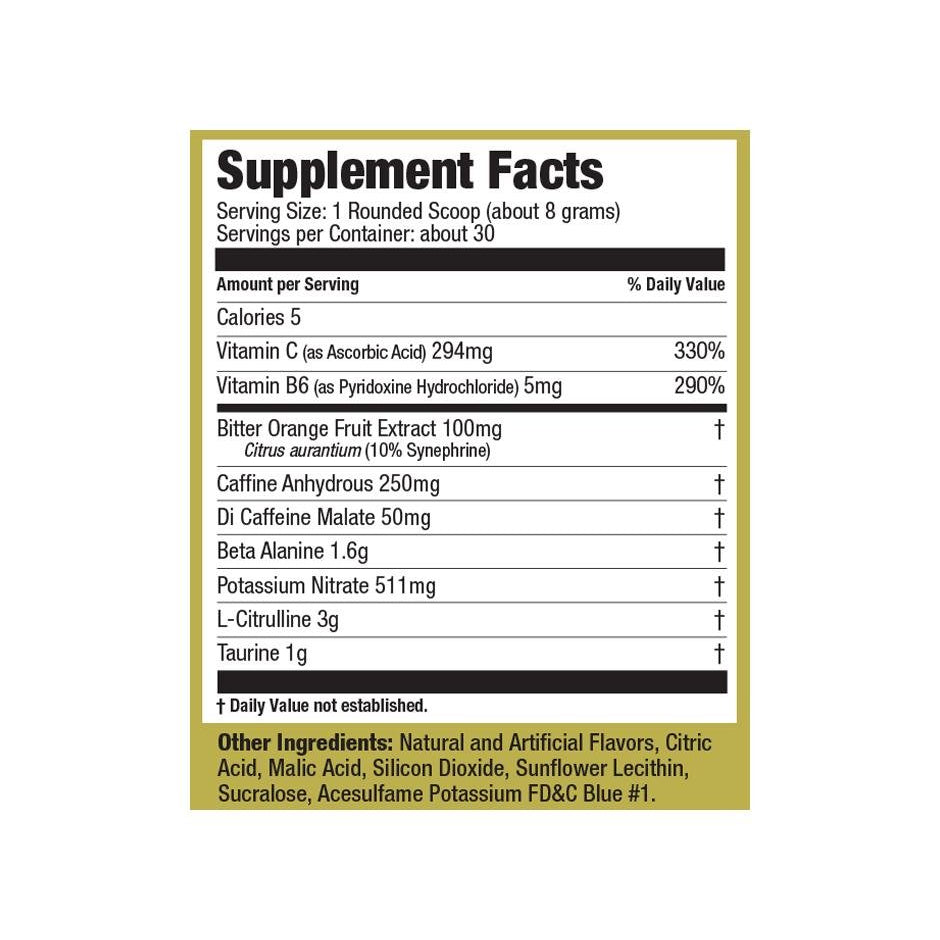
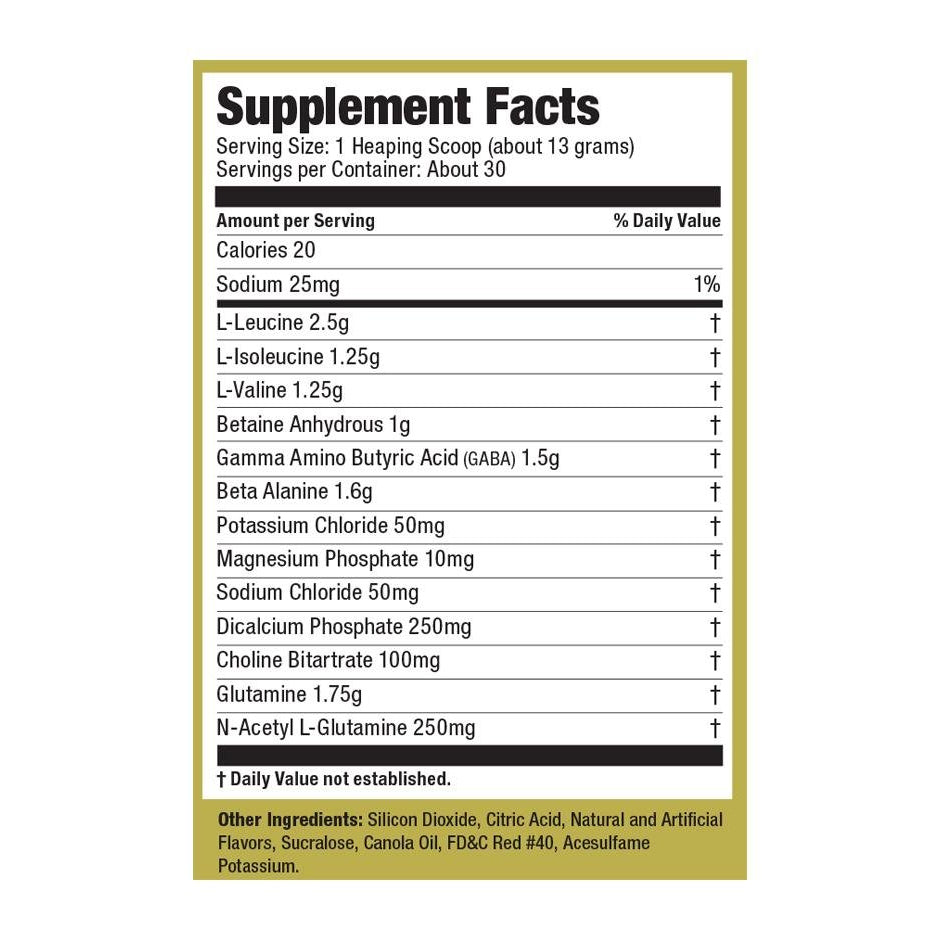
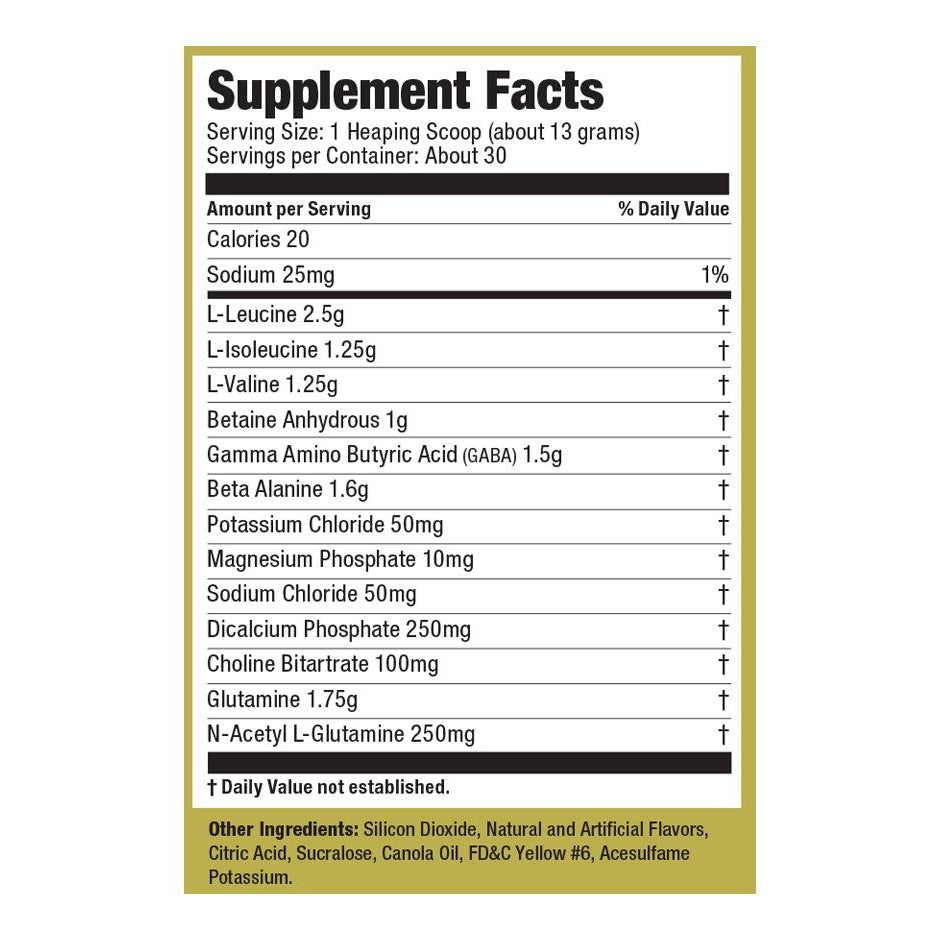
Explore Top-Selling Protein Powders
ISO SENSATION® 93
From
$33.99 USD
3.67 / 5.0
3 reviewsPROSTAR 100% WHEY PROTEIN
From
$24.99 USD
5.0 / 5.0
6 reviewsWHEY GOLD SAMPLE BOTTLE
$5.99 USD
1.0 / 5.0
1 reviewPushing Limits, Surpassing Goals
Over 40 Years of Innovation in Supplement Technology
Founded by Victor H. Rubino, a top powerlifter and visionary biochemist, Ultimate Nutrition began as a quest for superior supplements. Victor's passion for strength and science created a legacy of innovation in nutritional supplements.
From Our Community to Your Routine
Boost Workout Performance & Recovery
BCAA 12,000 POWDER
From
$21.99 USD
5.0 / 5.0
1 reviewTESTOSTROGROW® 2 HP
$49.99 USD
4.0 / 5.0
1 reviewHORSE POWER® X
$36.99 USD
5.0 / 5.0
1 reviewPre Gold: Unlock Your Performance Potential
$29.99 USD
5.0 / 5.0
1 reviewUP ALL NIGHT
$24.99 USD
5.0 / 5.0
1 reviewFree USA Shipping on orders $99+
Easy Returns on all orders
24/7 Customer support
FREE USA SHIPPING ON ORDERS $99+
our latest reviews
our latest reviews
Café Brazil is life changing!I don’t know where to start, this is perfect for summer I love coffee and you just have to add really cold water and you have iced coffee, it taste phenomenal, like I don’t know why I didn’t found you before and promise to be a loyal customer if you guys keep the same formula. It has pieces of coffee beans I think, that gives the extra kick, not too sweet, just awesome.
— S. Hernandez
our latest reviews
Amazing Product! Great flavor! Great company! Great product! I mixed the chocolate birthday cake protein powder with vanilla almond milk and I was blown away. It tastes exactly like a milkshake. It is not chalky or gritty at all. I would highly recommend this protein for before workouts, after workouts or just as a new replacement. PS, it has SPRINKLES in it.
— B. Emily
our latest reviews
Delicious with recipes! This product is actually very good on what it claims. I jazz it up w/ frozen banana, fresh strawberries, a tiny dash of sea salt & vanilla almond milk. the BEST strawberry shake w/o using ice cream. I love it! Curbs Hunger Definitely!! I'm finally able to not eat longer which helps with fasting. Love this product, I've recommended to several people
— N. Taylor
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON







 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON



 BUY OUR PRODUCTS ON
BUY OUR PRODUCTS ON




Pushing Limits, Surpassing Goals
Proven Excellence in High Quality Supplement Manufacturing

FDA Registered

ISO 17025 Compliant

HACCP Certified

GMP Facility
Must-Read Articles for Fitness Enthusiasts
Master the Cossack Squat: Benefits How-To and Progressions
The Cossack squat is a squat variation that can help you increase mobility and balance while strengthening your entire lower body. Learn why this all-around exercise needs to be in your training program.
—
UN Editorial Team
How Long Should You Work Out a Day?
Do you prefer quick workouts or long training sessions? In this guide, we'll uncover the truth about how long you should work out to get the most benefits.
—
UN Editorial Team
10 Deep Core Exercises to Build Abdominal Strength
Transform your mid-region with our top 10 deep core exercises for developing abdominal strength.
—
UN Editorial Team